Ernesto Lopes Pinheiro-Junior, Toxinologist Researcher, University of Leuven, Belgium.
Steve Peigneur, Postdoctoral Researcher, University of Leuven, Belgium.
Jan Tytgat, Full Professor, University of Leuven, Belgium.
 Animal venoms are known as incredible libraries of bioactive compounds acting on a plethora of different targets, with one basic objective: subduing and killing their preys. Composed mostly of peptides and larger proteins, toxins evolved to act on different targets such as endogenous substrates, second messengers, ion channels and G-protein coupled receptors (GPCRs). Several of these targets play a crucial role in the generation of action potentials and, consequently, in many others cellular activities like signal transduction, neurotransmitter release, muscular contraction, hormone secretion, cellular motility, cell differentiation, growth and apoptosis. In a nutshell, they are crucially involved in the generation of electrical signals, which propagate vital information across the organism.
Animal venoms are known as incredible libraries of bioactive compounds acting on a plethora of different targets, with one basic objective: subduing and killing their preys. Composed mostly of peptides and larger proteins, toxins evolved to act on different targets such as endogenous substrates, second messengers, ion channels and G-protein coupled receptors (GPCRs). Several of these targets play a crucial role in the generation of action potentials and, consequently, in many others cellular activities like signal transduction, neurotransmitter release, muscular contraction, hormone secretion, cellular motility, cell differentiation, growth and apoptosis. In a nutshell, they are crucially involved in the generation of electrical signals, which propagate vital information across the organism.
Given their importance in a myriad of physiological processes, it is not unexpected that many animal toxins target these structures. In this context, research encompassing ion channels and GPCRs has grown considerably in the last decades. Interestingly, most of the knowledge acquired about ion channels and GPRCs, involving the identification, structural and functional characterization, as well as their distribution throughout the human body was acquired thanks to the use of neurotoxins. The modulation of specific ion channels and GPCRs by animal toxins has triggered great interest since toxins can be considered prototypes for the development of novel compounds with potential therapeutic use. Additionally, it is now generally accepted that several ion channels are crucially involved in several channelopathies, giving rise to sometimes severe genetically related pathologies in humans. In this respect, our knowledge of ion channels and their structure-function is of pivotal importance.
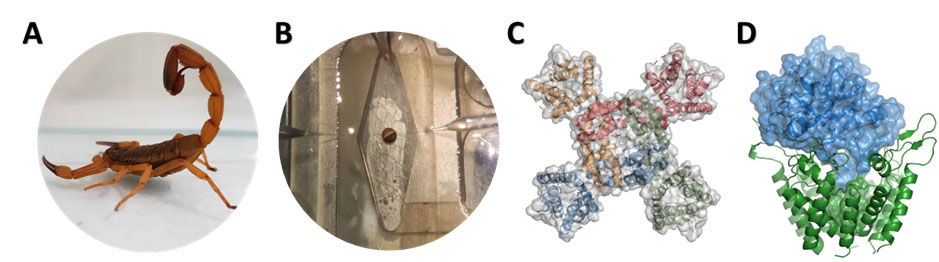
Image: Authors.
Figure 1. Schematic research flow in electrophysiology. (A) Tityus serrulatus specimens, from which venom toxins are purified. (B) Two-electrode voltage-clamp technique using Xenopus laevis oocytes. (C) Top view representation of a voltage-gated gated potassium channel (Kv). (D) Molecular docking of a toxin and a Kv channel. All images are from personal collections.
The research objectives of the Laboratory of Toxicology and Pharmacology, headed by Prof. Dr. Jan Tytgat at the University of Leuven (KU Leuven), Belgium – who is also part of the editorial board of JVATiTD – are mainly focused on the investigation of venom from animals such as spiders, sea anemones, cone snails, scorpions, cnidaria, etc. Since these venom toxins may act on ion channels and GPCRs throughout the human body, they are considered interesting lead compounds for the development of novel drugs to treat cancer, neurological disorders such as multiple sclerosis, Parkinson’s disease, or Alzheimer’s disease, as well as analgesics, insecticides, etc.
Currently, the research center holds a library of more than 80 different targets, comprising the cDNAs encoding different isoforms of voltage-gated potassium, sodium, and calcium channels, as well as ligand-gated ion channels and/or GPCRs, like nicotine-type acetylcholine, bradykinin, GABA-A, TRP, opioid, cannabinoid, histamine and bradykinin receptors. Through a wide network of collaborations with different partners across the globe, coupled with diverse assets and methodologies, we are key players in studying the interaction of different toxins with ion channels and GPCRs. The Laboratory of Toxicology and Pharmacology has particularly gained experience in the area of heterologous expression of those different cDNA targets. To this end, voltage-clamp experiments using Xenopus laevis oocytes are key in the bioassay used.
Different and interesting outcomes are being achieved through the use of the great potential animal toxins have as ion channel modulators. Recently, the group identified a toxin from a sea anemone capable of acting on 26 several voltage-gated ion channels, highlighting its remarkable promiscuity¹. On the other hand, a very selective and potent toxin towards a unique target was also identified², demonstrating the power of natural selection in picking powerful toxins, whose activities can be wisely employed in the development of novel therapeutics.
Given the impressive potential that toxins have in becoming remarkable drug leads, together with the urge in creating novel therapies for diseases without available treatment at the moment, the importance of identifying novel molecules and molecular targets becomes more than evident. This is exactly what moves us towards new goals and challenges in studying animal toxins: to find what is next and to discover what is beyond the frontiers of knowledge.
Read more
- PINHEIRO-JUNIOR, E.L., et al. A tale of toxin promiscuity: the versatile pharmacological effects of Hcr 1b-2 sea anemone peptide on voltage-gated ion channels. Marine Drugs [online]. 2022, vol. 20, no. 2, pp. 147 [viewed 7 June 2022]. https://doi.org/10.3390/md20020147. Available from: https://www.mdpi.com/1660-3397/20/2/147
- KUZMENKOV, A.I., et al. KV1.2 channel-specific blocker from Mesobuthus eupeus scorpion venom: structural basis of selectivity. Neuropharmacology [online]. 2018, vol. 143, pp. 228-238 [viewed 7 June 2022]. https://doi.org/10.1016/j.neuropharm.2018.09.030. Available from: https://doi.org/10.1016/j.neuropharm.2018.09.030
Links
Laboratory of Pharmacology and Toxicology: www.toxicology.be
Social Media – JVATITD: Facebook | Twitter
Journal of Venomous Animals and Toxins including Tropical Diseases: https://www.jvat.org/
Journal of Venomous Animals and Toxins including Tropical Diseases – JVATITD: https://www.scielo.br/j/jvatitd/
Como citar este post [ISO 690/2010]:

















Recent Comments