Daniela Barros, post graduate student of the Surgical Clinic Program at Ribeirão Preto Medical School, University of São Paulo (FMRP-USP). Ribeirão Preto, SP, Brasil.

A group of researchers from Faculdade de Ciências Médicas da Santa Casa de São Paulo (São Paulo, SP, Brazil), conducted a retrospective study of 517 patients with thyroid microcarcinoma (TMC) treated with total thyroidectomy, with or without radioactive iodine (RAI) therapy, reclassified after 1.1 ± 0.4 years according to the response to treatment into “favorable” (excellent/indeterminate) or “unfavorable” (biochemical/ structural incomplete) responses. Named “Prognostic factors for incomplete response in thyroid microcarcinoma: an analysis of initial response to therapy in 517 patients”, the article evaluated participants’ age, sex, tumor size, histological variants, multifocality, presence of vascular/lymphatic/perineural invasion, extrathyroidal extension, metastatic lymph nodes (LN), and distant metastasis (FARO, F.N, et al).
Thyroid cancer is the most common endocrine neoplasm, and its incidence is on a significant rise in the last 30 years (HOWLADER, N. et al). Moreover, mortality from thyroid cancer remains low and stable, suggesting that TMC is, in most cases, an indolent tumor, detected incidentally on ultrasound examination (AHN, H.S., et al; LI, M., et al). Therefore, with its low risk of persistence and/or recurrence and a mortality rate of less than 1% (MAZZAFERRI, E.L.), active surveillance has been proposed as an appropriate therapeutic strategy for low-risk patients, with outcomes like to that of surgical treatment (MIYAUCHI, A.).
Most studies involving risk factors for TMC focus on parameters that predispose to the development of lymph node (LN) metastasis at diagnosis (LUO, Y., et al; HUANG, X.P., et al; KALISZEWSKI, K., et al; GUI, C.Y. et al). However, according to the guidelines of the American Thyroid Association (ATA) 2015 (HAUGEN, B.R. et al), a good way to assess a patient’s prognosis is to classify them correctly according to the risk of recurrence or persistence of the disease and on the type of response after 6–24 months of the initial treatment (TUTTLE, R.M., et al), considering not only evidence of structural disease but also the values of serum thyroglobulin (Tg) and anti-Tg antibody (TgAb) levels.
Image: peoplecreations
The main goal of this study, led by Faro and cols., was to identify possible risk factors associated with an initial incomplete response in patients with TMC. They observed that multifocality and LN metastasis are independent risk factors for incomplete response in TMC patients and are strongly correlated. Additional RAI therapy was not associated with a more favorable response in these subgroups (FARO, F.N, et al).
A total of 5344 patients who underwent thyroid surgery for malignancy between 1972 and 2015 compose the database of DTC. Patients who were diagnosed with TMC (a total of 1840) were selected. TMC was defined as a thyroid tumor 1 cm or less in diameter, in accordance with the WHO classification (DE LELLIS, R. et al) Subsequently, the available information allowed to classify 517 patients according to the ATA dynamic risk stratification using the response to therapy restaging system (HAUGEN, B.R. et al). The patients were evaluated retrospectively.
The patients were reevaluated 6-24 months after initial treatment by measuring their levels of thyroid-stimulating hormone (TSH), Tg, TgAb and stimulated thyroglobulin (sTg), and by performing a neck US, with or without a WBS. Overall, 125 patients did not undergo sTg. In these cases, the suppressed Tg level was used as an alternative.
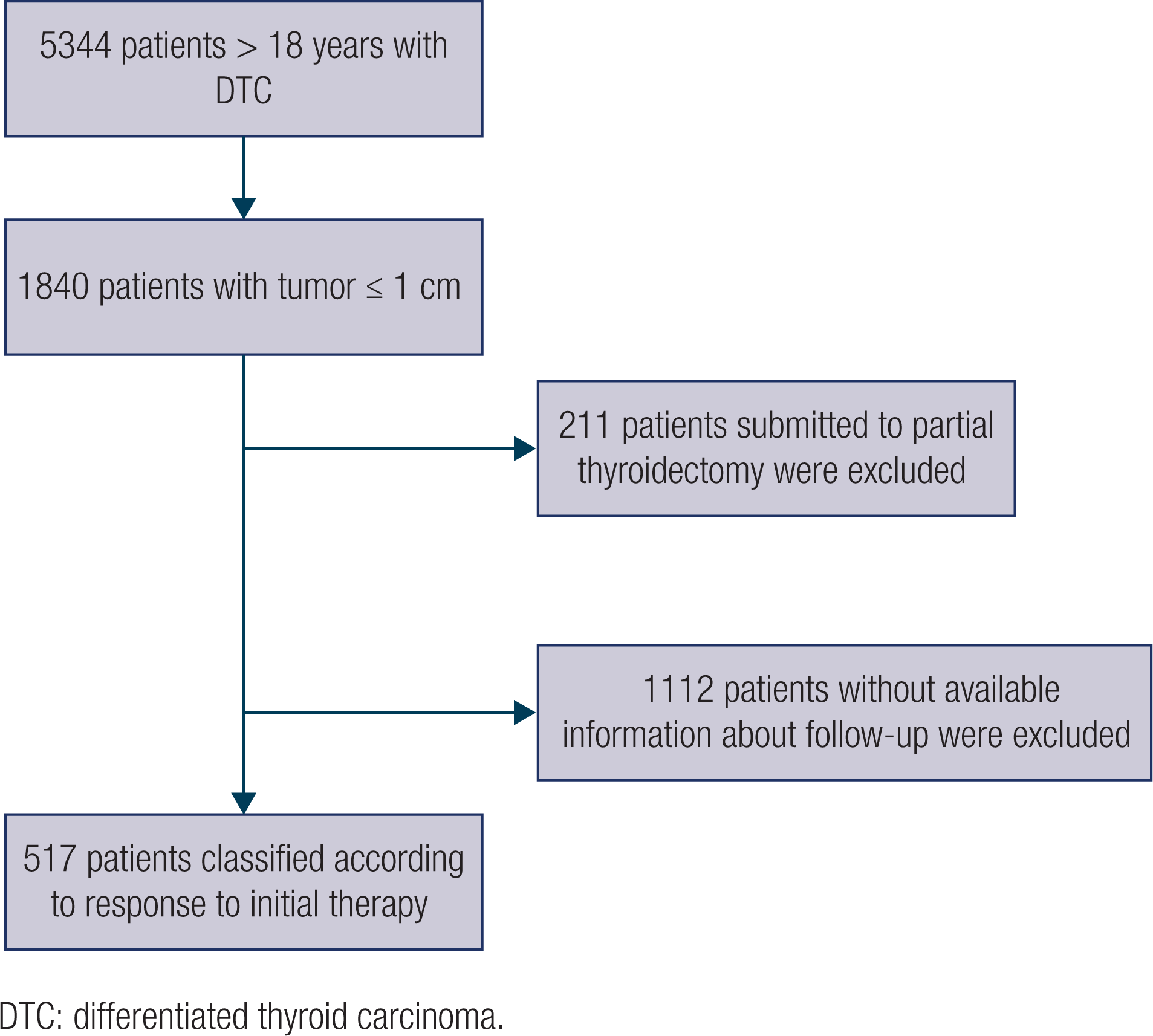
Image: FARO, F.N., et al.
Due to the evidence that TMC generally has an indolent course and a low risk of recurrence and/or persistence, the ATA considers the possibility of active surveillance instead of immediate surgery in patients with low-risk TMC (HAUGEN, B.R. et al). However, the identification of prognostic factors is important in deciding which patients are more suitable for active surveillance and for other therapeutic strategies.
In accordance with Ito and cols. (ITO, Y. et al) and Tuttle and cols. (TUTTLE, R.M., et al), patients with lymph node (LN) involvement or signs of extrathyroidal extension are not eligible for active surveillance. Moreover, Brito and cols. (BRITO, J.P., et al) divided patients into ideal, appropriate, and inappropriate candidates for active surveillance. In these categories, the authors suggest that the presence of extrathyroidal extension, multifocality LN metastasis and other factors should be considered.
This is the first study to correlate risk factors with the response range of dynamic ATA risk stratification in patients with TMC, where 20.7% of the subjects with TMC presented with an unfavorable response (biochemical and structural incomplete responses) after the initial treatment. This is higher than that reported by Mazzaferri and cols. (MAZZAFERRI, E.L.), who found locoregional and distant recurrences of TMC in 5.9% and 1.5%, respectively.
Therefore, the authors conclude that although patients with TMC are considered low risk and evolve well, there is a particular group of patients which can evolve with an incomplete response and, thereby, benefit from surgical treatment without complementary radioiodine treatment, as the latter did not show any additional benefit for this group of patients in our study. Long-term follow-up studies are strongly recommended to corroborate these findings.
References
AHN, H.S., et al. Korea’s thyroid-cancer “epidemic” — screening and overdiagnosis. New England Journal of Medicine [online]. 2014, vol. 371, no 19, pp. 1765-7. [viewed 16 Nov 2021]. https://doi.org/10.1056/nejmp1409841. Available from: https://www.nejm.org/doi/10.1056/NEJMp1409841?url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org&rfr_dat=cr_pub++0pubmed
BRITO, J.P., et al. A clinical framework to facilitate risk stratification when considering an active surveillance alternative to immediate biopsy and surgery in papillary microcarcinoma. Thyroid [online]. 2016, vol. 26, no. 1, pp. 144-9. [viewed 16 Nov 2021]. https://doi.org/10.1089/thy.2015.0178. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4842944/
DE LELLIS, R., et al. World Health Organization classification of tumours: pathology and genetics of tumours of the endocrine organs. Lyon: International Agency for Research on Cancer (IARC), 2004.
GUI, C.Y., et al. Clinical and pathologic predictors of central lymph node metastasis in papillary thyroid microcarcinoma: a retrospective cohort study. Journal of Endocrinological Investigation [online]. 2018, vol. 41, no. 4, pp. 403-9. [viewed 16 Nov 2021]. https://doi.org/10.1007/s40618-017-0759-y. Available from: https://link.springer.com/article/10.1007/s40618-017-0759-y
HAUGEN, B.R., et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid [online]. 2016, vol. 26, no. 1, pp.1-133. [viewed 16 Nov 2021]. https://doi.org/10.1089/thy.2015.0020. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4739132/
HOWLADER, N., et al.SEER Cancer Statistics Review, 1975–2016 [online]. National Cancer Institute, 2019 [viewed 8 December 2021]. Available from: https://seer.cancer.gov/csr/1975_2016/
HUANG, X.P., et al. Sonographic features of papillary thyroid microcarcinoma predicting high-volume central neck lymph node metastasis. Surgical Oncology [online]. 2018, vol. 27, no. 2, pp.172-6. [viewed 16 Nov 2021]. https://doi.org/10.1016/j.suronc.2018.03.004. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0960-7404(17)30408-5
ITO, Y., et al. Active surveillance of low-risk papillary thyroid microcarcinomas in Japan and other countries: a review. Expert Review of Endocrinology & Metabolism [online]. 2020, vol. 15, no. 1, pp. 5-12. [viewed 16 Nov 2021]. https://doi.org/10.1080/17446651.2020.1707078 Available from: https://pubmed.ncbi.nlm.nih.gov/31902250/
KALISZEWSKI, K., et al. Which papillary thyroid microcarcinoma should be treated as “true cancer” and which as “precancer”? World Journal of Surgical Oncology [online]. 2019, vol. 17, no. 1, pp. 91. [viewed 16 Nov 2021]. https://doi.org/10.1186/s12957-019-1638-0. Available from: https://wjso.biomedcentral.com/articles/10.1186/s12957-019-1638-0
LI, M., et al. Global trends in thyroid cancer incidence and the impact of overdiagnosis. Lancet Diabetes Endocrinol [online]. 2020, vol. 8, no. 6, pp.468-70. [viewed 16 Nov 2021]. https://doi.org/10.1016/S2213-8587(20)30115-7. Available from: https://www.thelancet.com/journals/landia/article/PIIS2213-8587(20)30115-7/fulltext
LUO, Y., et al. Clinical analysis of cervical lymph node metastasis risk factors in patients with papillary thyroid microcarcinoma. Journal of Endocrinological Investigation [online]. 2019, vol. 42, no. 2, pp. 227-36. [viewed 16 Nov 2021]. https://doi.org/10.1007/s40618-018-0908-y. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6394766/
MAZZAFERRI, E.L. Management of low-risk differentiated thyroid cancer. Endocrine Practice [online]. 2007, vol. 13, no. 5, pp. 498-512. [viewed 16 Nov 2021]. https://doi.org/10.4158/EP.13.5.498. Available from: https://www.sciencedirect.com/science/article/abs/pii/S1530891X20420452
MIYAUCHI, A. Clinical trials of active surveillance of papillary microcarcinoma of the thyroid. World Journal of Surgery [online]. 2016, vol. 40, no. 3, pp.516-22. [viewed 16 Nov 2021]. https://doi.org/10.1007/s00268-015-3392-y. Available from: https://link.springer.com/article/10.1007%2Fs00268-015-3392-y
TUTTLE, R.M., et al. A clinical framework to facilitate selection of patients with differentiated thyroid cancer for active surveillance or less aggressive initial surgical management. Expert Review of Endocrinology & Metabolism [online]. 2018, vol. 13, no. 2, pp.77-85. [viewed 16 Nov 2021]. https://doi.org/10.1080/17446651.2018.1449641. Available from: https://pubmed.ncbi.nlm.nih.gov/26462967/
TUTTLE, R.M., et al. Estimating risk of recurrence in differentiated thyroid cancer after total thyroidectomy and radioactive iodine remnant ablation: using response to therapy variables to modify the initial risk estimates predicted by the new American Thyroid Association staging system. Thyroid [online]. 2010, vol. 20, no. 12, pp. 1341-9. [viewed 16 Nov 2021]. https://doi.org/10.1089/thy.2010.0178. Available from: https://pubmed.ncbi.nlm.nih.gov/26462967/
External Links
Fernanda Nascimento Faro: http://orcid.org/0000-0002-3240-075X
Archives of Endocrinology and Metabolism – AEM: https://www.scielo.br/aem
To read the article, acess
FARO, F.N., et al. Prognostic factors for incomplete response in thyroid microcarcinoma: an analysis of initial response to therapy in 517 patients. Archives of Endocrinology and Metabolism [online]. 2021, vol. 65, no. 5, pp. 579-87. [viewed 16 Nov 2021]. https://doi.org/10.20945/2359-3997000000341. Available from: https://www.scielo.br/j/aem/a/y4rbrJmHVZbnkDTm4WG3Jxx/?lang=en#
Como citar este post [ISO 690/2010]:















Recent Comments