Daniela Barros, post graduate student of the Surgical Clinic Program at Ribeirão Preto Medical School, University of São Paulo (FMRP-USP). Ribeirão Preto, SP, Brasil.
 Transforming growth factor-β1 (TGF-β1) is a multifunctional cytokine that plays a role in critical functions such as cellular differentiation, migration, apoptosis, and regulation of the immune systems (CHIN, D., et al). Abnormalities involving the TGFB1 gene and its receptors are common in several types of cancer, including thyroid malignancy, and often related to tumor progression.
Transforming growth factor-β1 (TGF-β1) is a multifunctional cytokine that plays a role in critical functions such as cellular differentiation, migration, apoptosis, and regulation of the immune systems (CHIN, D., et al). Abnormalities involving the TGFB1 gene and its receptors are common in several types of cancer, including thyroid malignancy, and often related to tumor progression.
Single nucleotide polymorphisms (SNP) are genetic variations often distributed throughout the human genome, and their location can interfere in different biological processes. Easily accessed nowadays, these SNPs can provide valuable information by identifying individuals genetically susceptible to multifactorial diseases, the aggressiveness of the disease, and poor response to treatments.
In order to better understand their role in the susceptibility and clinical features of thyroid cancer, Peres e cols. analyzed some TGFB1, TGFBR1, and TGFBR2 SNPs previously associated with human cancers as well as SNPs that have been implicated on gene and protein deregulation (MARTELOSSI, C., et al; LIU, J., et al; HE, B., et al; IKEDA, H., et al).
In an article called “Clinical utility of TGFB1 and its receptors (TGFBR1 and TGFBR2) in thyroid nodules: evaluation based on single nucleotide polymorphisms and mRNA analysis” (Arch Endocrinol Metab. 2021;65(2):172-84), the authors genotyped TGFB1, TGFBR1, and TGFBR2 SNPs in 157 papillary thyroid cancer (PTC) patients and 200 healthy controls. Further, they investigated RNA samples of 47 PTC and 80 benign nodules, searching for differential mRNA expression. They found out that TGFB1 polymorphism rs1800472 may confer greater activity to TGF-β1 in the tumor microenvironment, favoring PTC aggressiveness.
Although most guidelines restrict the indication for further investigation of small thyroid nodules and the criteria for malignancy have become stricter, an increasing number of patients end up referred to fine-needle aspiration (FNA) biopsy for diagnostic confirmation and many are submitted to surgery. In this scenario, it is fundamental to find ways to optimize the management of these patients, avoiding inappropriate and excessive spending on the health system, besides ensuring patients’ physical and psychological well-being (DURANTE, C., et al). This is one of the main reasons why a collaboration between researchers from Universidade Estadual de Campinas (Brazil) and Universidade do Porto, Porto, Portugal was established.
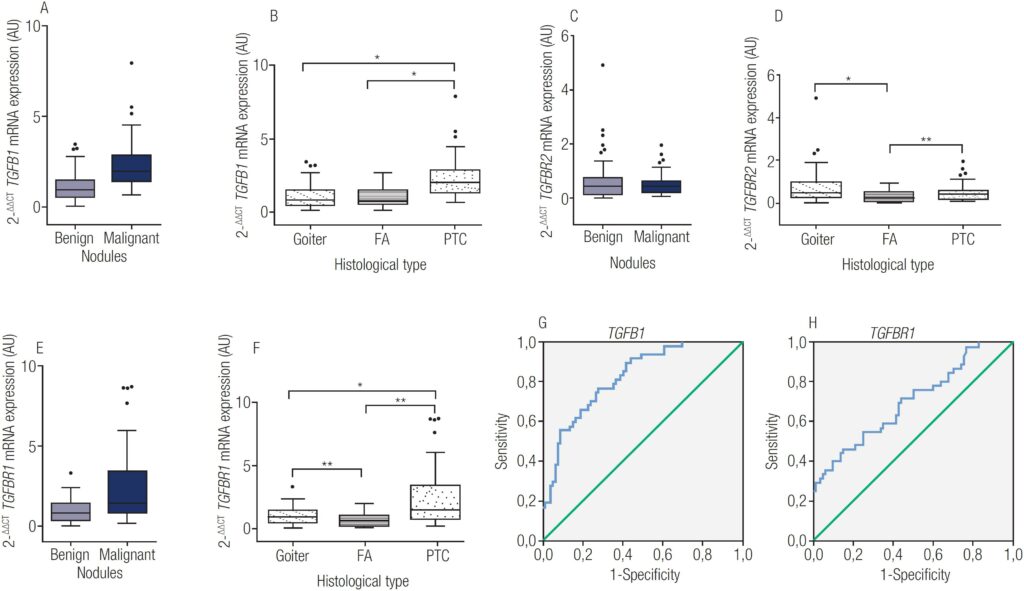
transforming growth factor-β1 (TGFB1) mRNA expression in 80 benign and 47 PTC, p<0.0001. B: TGFB1 mRNA expression in the different types of tissues analyzed, *all comparisons p<0.0001. C: transforming growth factor receptor I (TGFBR1) mRNA expression in 80 benign and 47 PTC, p<0.0001. D: TGFBR1 mRNA expression in the different types of tissues analyzed, *PTC versus goiter – p=0.0049, **PTC versus FA – p<0.0001, ***goiter versus FA – p=0.0267. E: transforming growth factor receptor II (TGFBR2) mRNA expression in 80 benign and 47 PTC, p-value not significant. F: TGFBR2 mRNA expression in the different types of tissues analyzed, *goiter versus FA – p=0.0002, **PTC versus FA – p=0.0120. G: ROC analysis for TGFB1 using a cut-off point of 1.365: sensitivity of 77%, specificity of 72%, PPV of 12%, NPV of 98%, and an area under the curve (AUC) of 0.821 (p<0.0001). H: ROC analysis for TGFBR1 with sensitivity of 47%, specificity of 85%, PPV of 14%, NPV of 97%, and AUC of 0.701 (p=0.0002). Image: PERES, K. C., et al.
The team identified that SNPs rs1800472 and rs1800469 were associated with characteristics of PTC aggressiveness. Effect predictor software analysis of nonsynonymous SNP rs1800472 indicated increasing protein stability and post-translational changes. TGFB1 mRNA expression was upregulated in PTC and downregulated in benign samples (follicular adenomas; FA), differentiating malignant from benign nodules (p<0.0001); PTC from goiter (p<0.0001); and PTC from FA (p<0.0001). TGFBR1 mRNA expression was upregulated in goiter and PTC, but downregulated in FA, distinguishing PTC from goiter (p=0.0049); PTC from FA (p<0.0001); and goiter from FA (p=0.0267). On the other hand, TGFBR2 was downregulated in all histological types analyzed and was not able to differentiate thyroid nodules.
Peres e cols. concluded that some polymorphisms, such as rs1800472, may modulate TGF-β1 activity and help define PTC aggressiveness. Therefore, evaluating TGFB1 and TGFBR1 mRNA levels may be useful to characterize thyroid nodules malignancy.
References
CHIN, D., et al. What is transforming growth factor-beta (TGF-beta)? Br J Plast Surg [online]. 2004, vol. 57, nº 3, pp.215-221 [viewed 05 May 2021]. https://doi.org/10.1016/j.bjps.2003.12.012. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0007122603005861
MARTELOSSI, C., et al. TGF-β1 functional polymorphisms: a review. Eur Cytokine Netw [online]. 2016, vol. 27, nº 4, pp.81-89 [viewed 05 May 2021]. https://doi.org/10.1684/ecn.2016.0382. Available from: https://www.jle.com/fr/revues/ecn/e-docs/tgf_1_functional_polymorphisms_a_review_309493/article.phtml
LIU, J., et al. Genetic polymorphism contributes to 131I radiotherapy-induced toxicities in patients with differentiated thyroid cancer. Pharmacogenomics [online]. 2018, vol. 19, nº 17, pp.1335-1344 [viewed 05 May 2021]. https://doi.org/10.2217/pgs-2018-0070. Available from: https://www.futuremedicine.com/doi/abs/10.2217/pgs-2018-0070
HE, B., et al. Polymorphisms of TGFBR1, TLR4 are associated with prognosis of gastric cancer in a Chinese population. Cancer Cell Int. 2018, vol. 18, pp.191 [viewed 05 May 2021]. https://doi.org/10.1186/s12935-018-0682-0. Available from: https://cancerci.biomedcentral.com/articles/10.1186/s12935-018-0682-0
IKEDA, H. Mutational analysis of transforming growth factor-beta receptor type II and Smad3 tumor suppressor genes in prolactinomas. Brain Tumor Pathol [online]. 2006, vol. 23, nº 1, pp. 7-12. [viewed 05 May 2021]. https://doi.org/10.1007/s10014-006-0196-7. Available from: https://link.springer.com/article/10.1007/s10014-006-0196-7
DURANTE, C., et al. The Diagnosis and Management of Thyroid Nodules: A Review. JAMA [online]. 2018, vol. 319, nº 9, pp. 914-924 [viewed 05 May 2021]. https://doi.org/10.1001/jama.2018.0898. Available from: https://jamanetwork.com/journals/jama/article-abstract/2673975
To read the article, acess
PERES, K. C., et al. Clinical utility of TGFB1 and its receptors (TGFBR1 and TGFBR2) in thyroid nodules: evaluation based on single nucleotide polymorphisms and mRNA analysis. Arch Endocrinol Metab [online]. 2021, vol. 65, nº 2, pp.172-184 [viewed 05 May 2021]. https://doi.org/10.20945/2359-3997000000330. Available from: http://ref.scielo.org/kmp7vf
External links
Archives of Endocrinology and Metabolism – AEM: https://www.scielo.br/aem
Karina Colombera Peres: https://orcid.org/0000v-0003-3253-371X
Como citar este post [ISO 690/2010]:
















Recent Comments